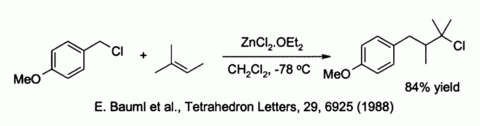
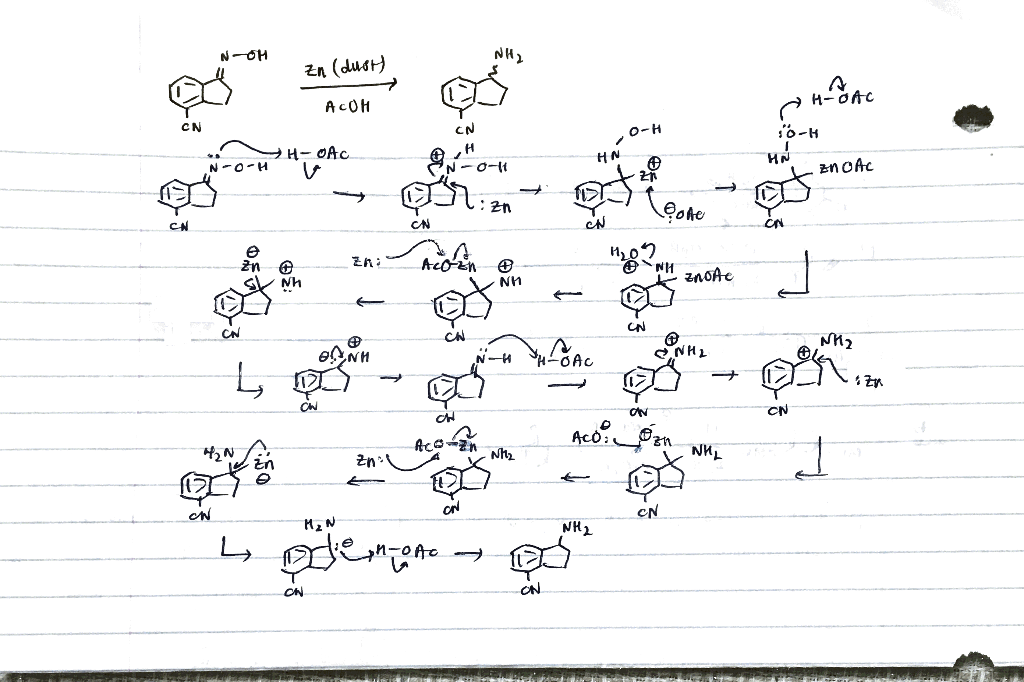
A FACILE AND EFFICIENT SYNTHESIS OF HIGHLY FUNCTIONALIZED TERMINAL OLEFINES FROM á-ALKOXY-ß-HALIDES USING ZINC DUSTΨ
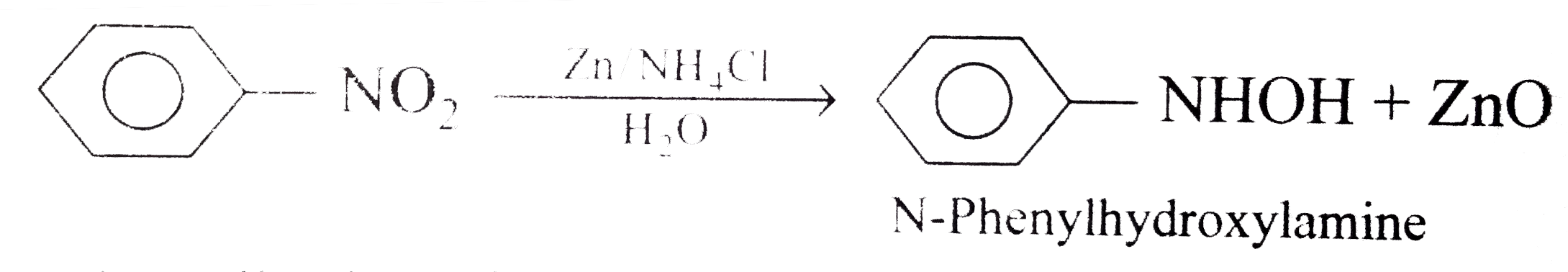
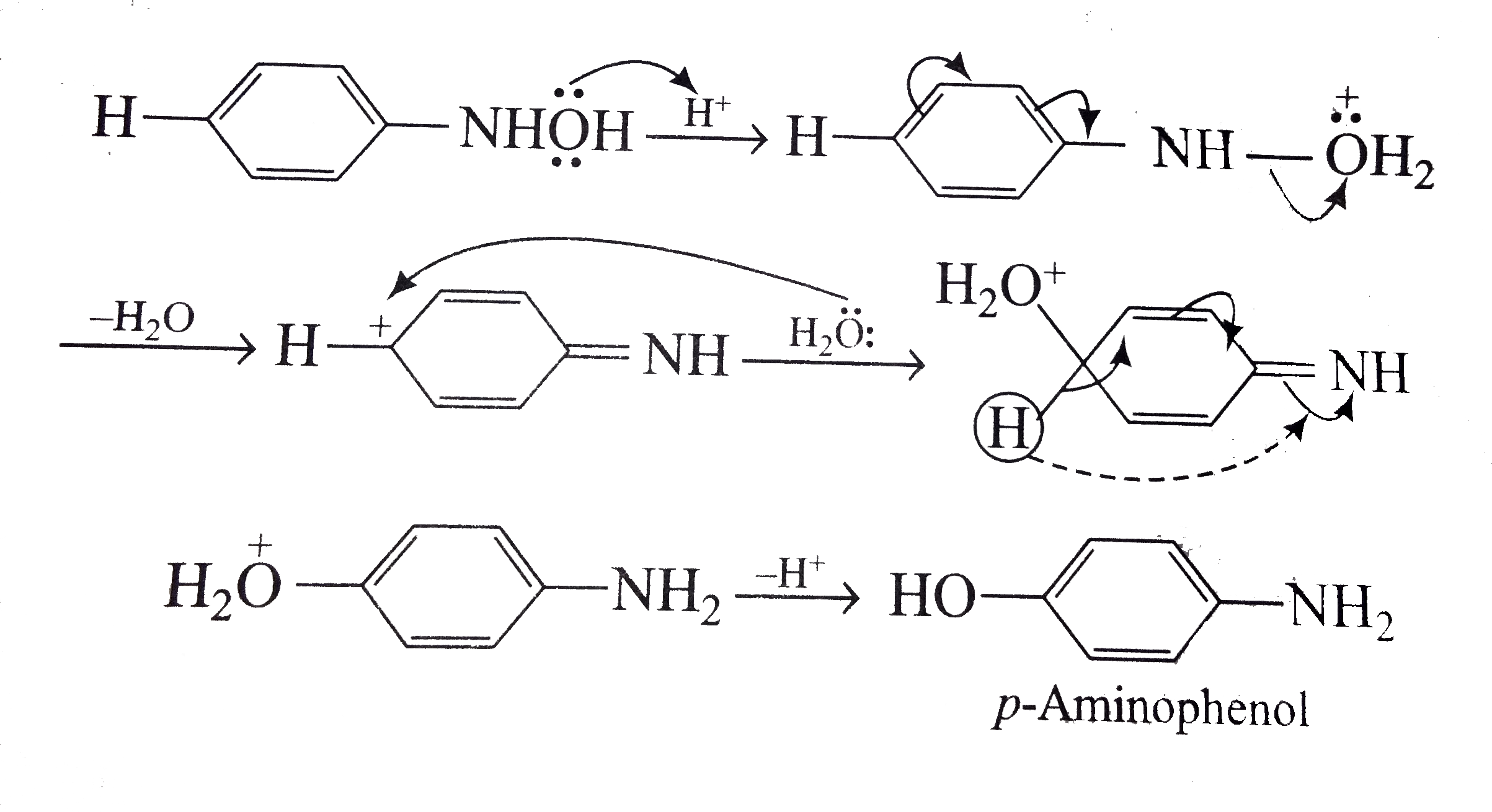
Sciencemadness Discussion Board - Reduction of nitrobenzene by zinc and ammonium chloride - Powered by XMB 1.9.11

Reduction of nitrobenzene with zinc dust and aqueous ammonium chloride yields | 12 | AMINES | C... - YouTube
Sciencemadness Discussion Board - Reduction of nitrobenzene by zinc and ammonium chloride - Powered by XMB 1.9.11

Reduction of aromatic nitro compounds to amines using zinc and aqueous chelating ethers: Mild and efficient method for zinc activation | Semantic Scholar

A FACILE AND EFFICIENT SYNTHESIS OF HIGHLY FUNCTIONALIZED TERMINAL OLEFINES FROM á-ALKOXY-ß-HALIDES USING ZINC DUSTΨ
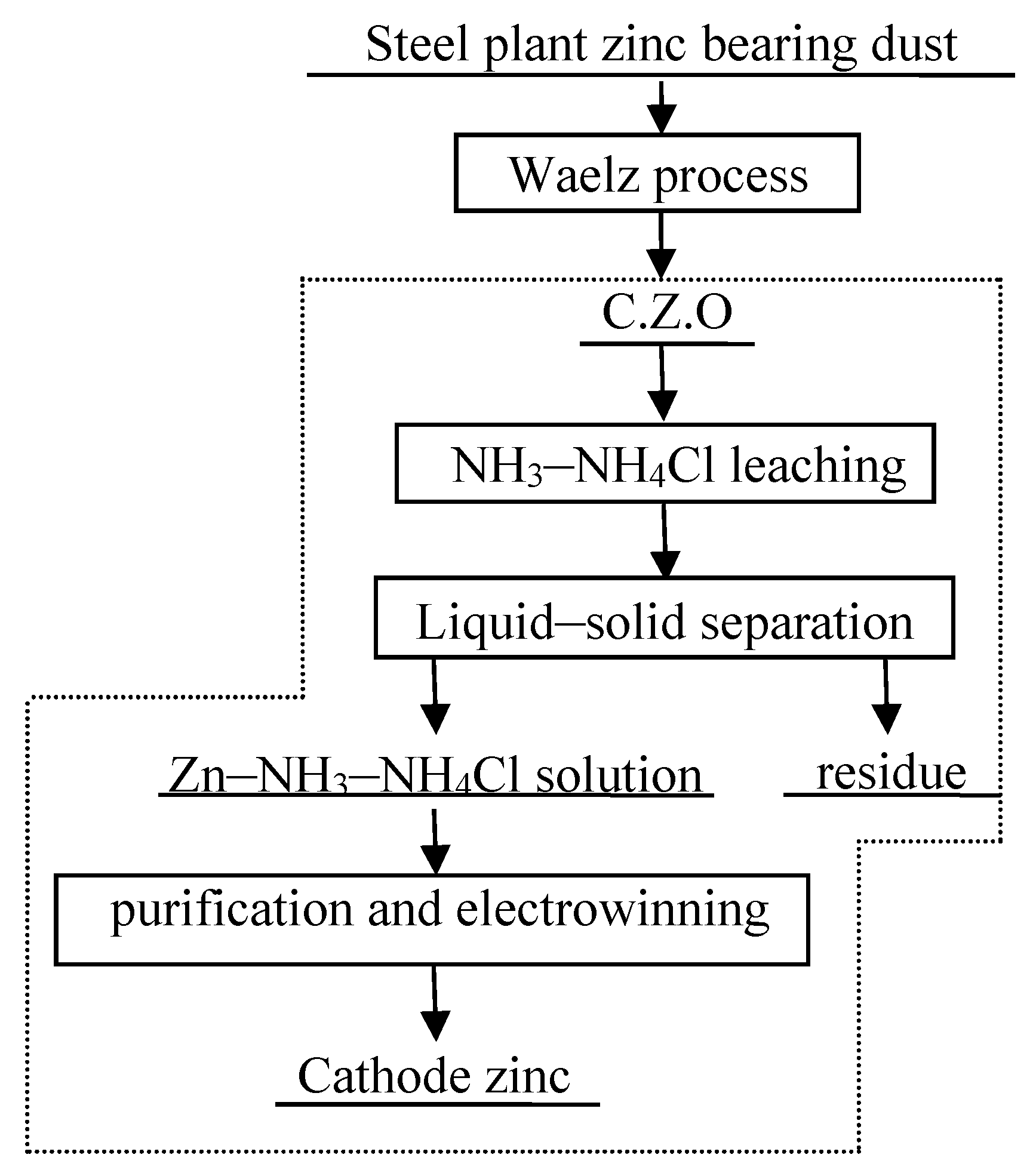
Metals | Free Full-Text | Hydrometallurgical Process for Zinc Recovery from C.Z.O. Generated by the Steelmaking Industry with Ammonia–Ammonium Chloride Solution | HTML

Sciencemadness Discussion Board - Reduction of nitrobenzene by zinc and ammonium chloride - Powered by XMB 1.9.11
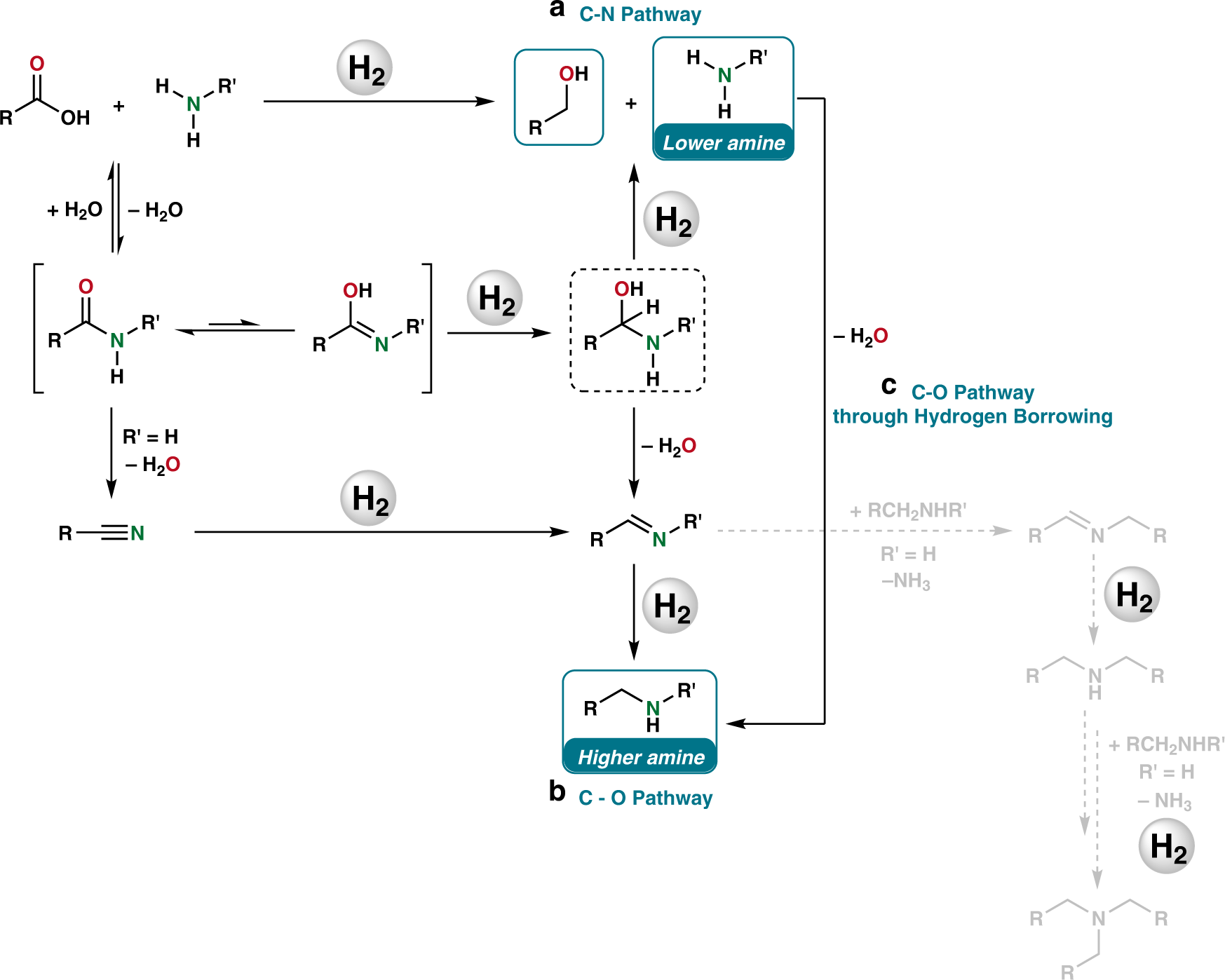
Homogeneous and heterogeneous catalytic reduction of amides and related compounds using molecular hydrogen | Nature Communications

NH4Cl/Zn powder: An efficient, chemoselective reducing catalyst for the microwave‐assisted synthesis of 2,3‐disubstituted quinolines via tandem Knoevenagel condensation - Tasqeeruddin - 2021 - Journal of Heterocyclic Chemistry - Wiley Online Library