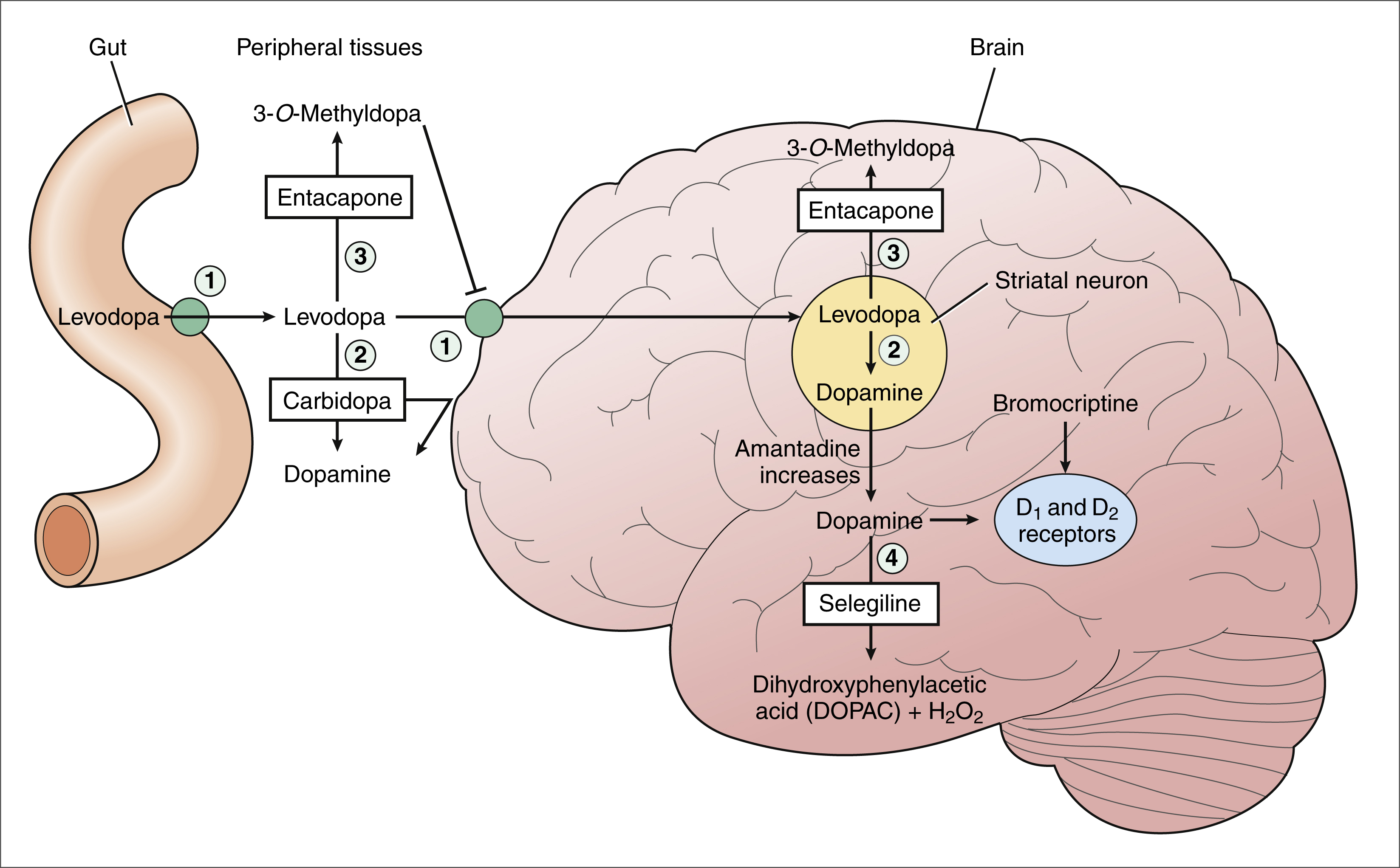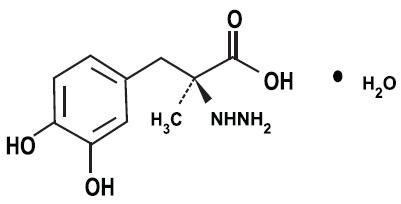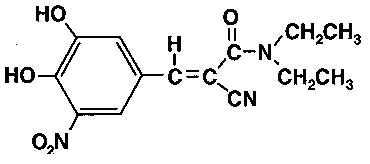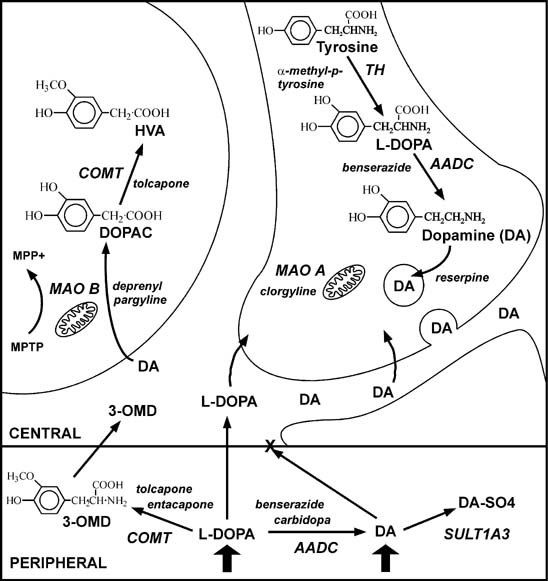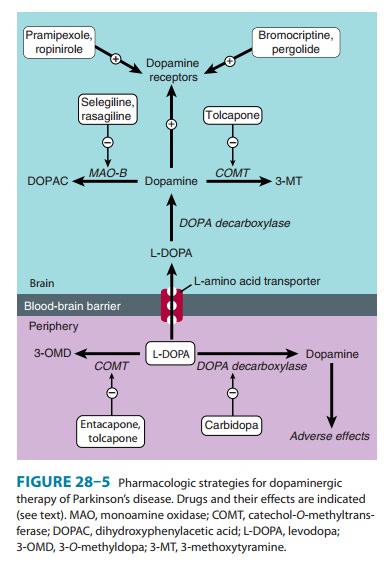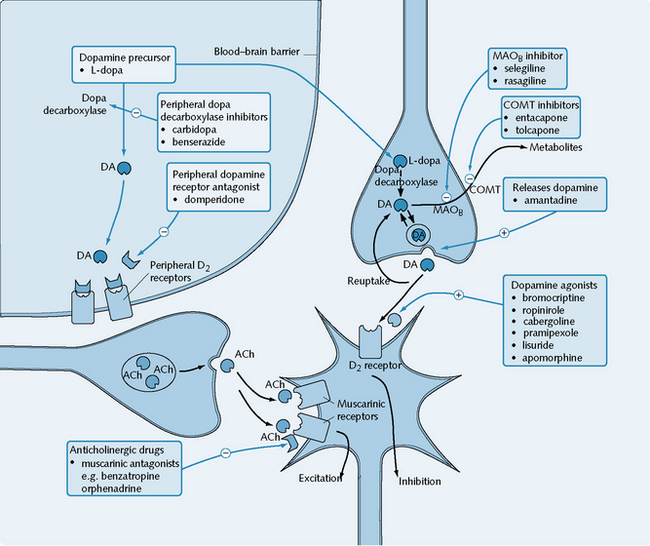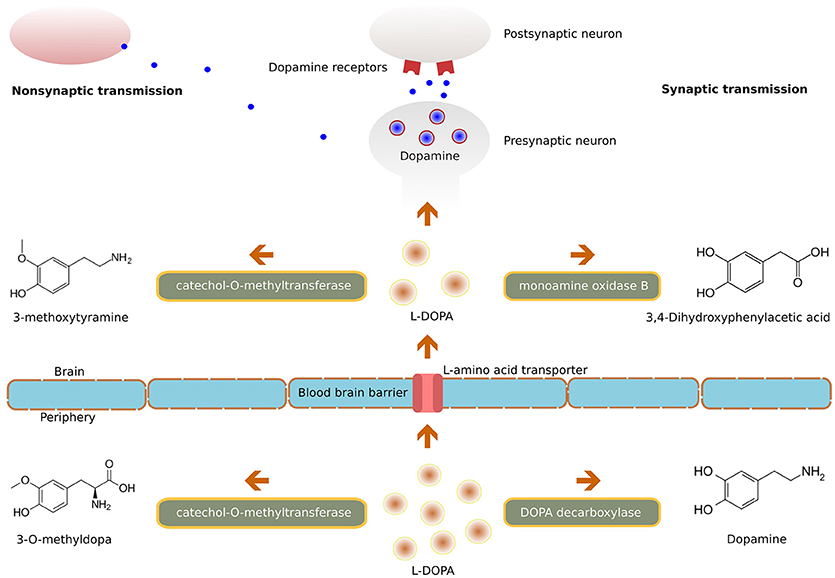
Frontiers | Deep Brain Stimulation and L-DOPA Therapy: Concepts of Action and Clinical Applications in Parkinson's Disease | Neurology

Relating mode of action to clinical practice: Dopaminergic agents in Parkinson's disease - ScienceDirect
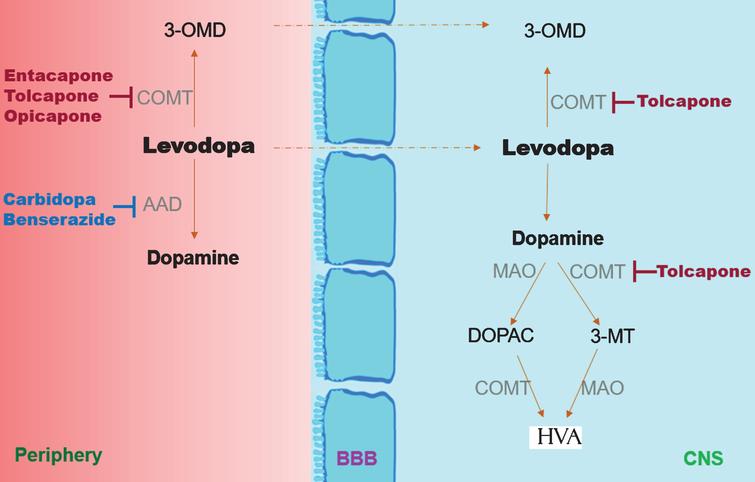
Are There Benefits in Adding Catechol-O Methyltransferase Inhibitors in the Pharmacotherapy of Parkinson's Disease Patients? A Systematic Review - IOS Press

Entacapone and Tolcapone, Two Catechol O-Methyltransferase Inhibitors, Block Fibril Formation of α-Synuclein and β-Amyloid and Protect against Amyloid-induced Toxicity - ScienceDirect
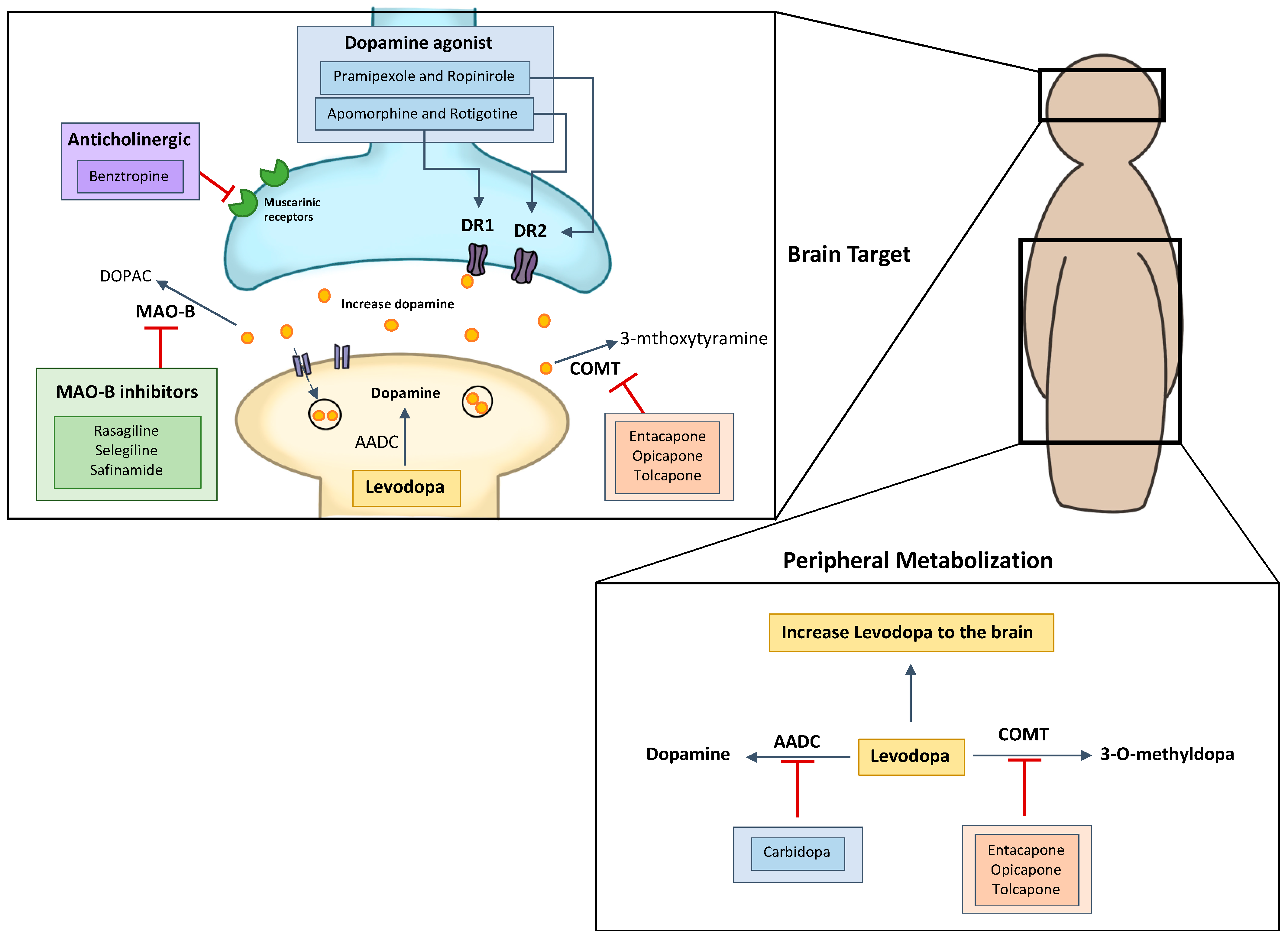
Pharmaceutics | Free Full-Text | Importance of Nanoparticles for the Delivery of Antiparkinsonian Drugs | HTML

Advances and Challenges in the Dosage Form Design for the Treatment of Parkinson's Disease - Ming Ming Wen - Discovery Medicine
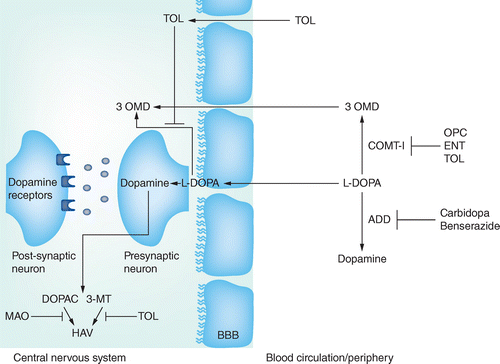
Opicapone for Parkinson's disease: clinical evidence and future perspectives | Neurodegenerative Disease Management